Various Dry Shampoos
Allegations: Failing to disclose products contain the carcinogen benzene
Poppiti et al. v. Rite Aid Corp.
23-cv-3914, S.D.N.Y.
(May 2023)
Rite Aid Oral Care Dry Mouth Discs
Misleadingly marketing that products “promote a healthy mouth,” “provide lasting moisture,” and “soothe[] dry tissues” when using them contributes to various oral health issues, including dental erosion and sensitivity
Pending
Allegations: Failing to disclose products contain the carcinogen benzene
Allegations: Misrepresenting that it protects consumers’ personal information when it failed to do so and there was a data breach in June 2024
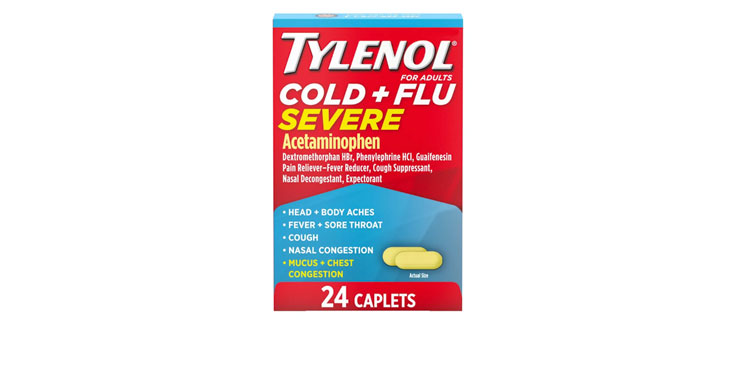
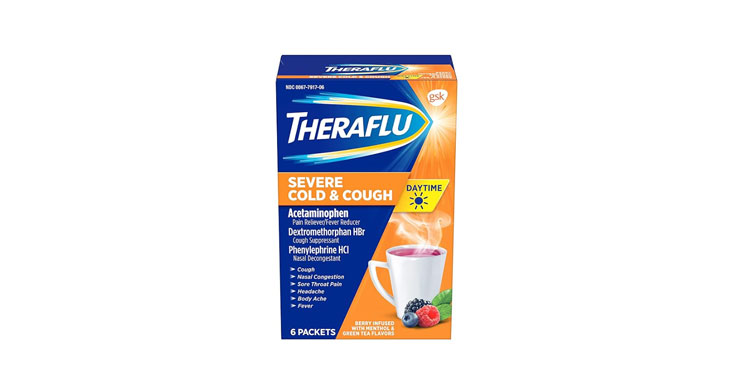
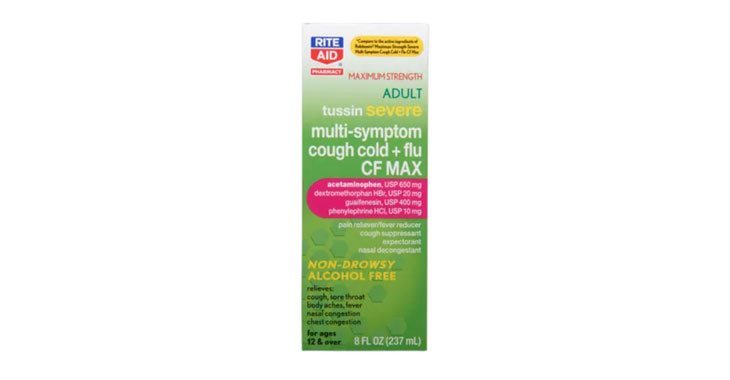
Allegations: Falsely marketing that phenylephrine products treat congestion and other cold and flu symptoms
Allegations: Falsely marketing that medicines treat nasal congestion
Allegations: Falsely marketing medicines as “Non-Drowsy” when the active ingredient causes drowsiness
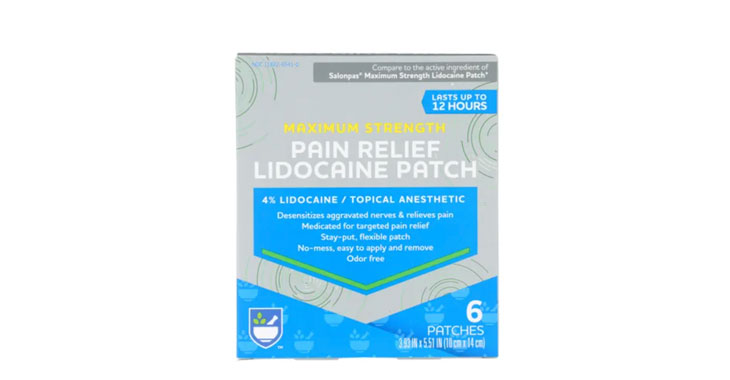
Allegations: Misleadingly marketing Rite Aid pain relief patches
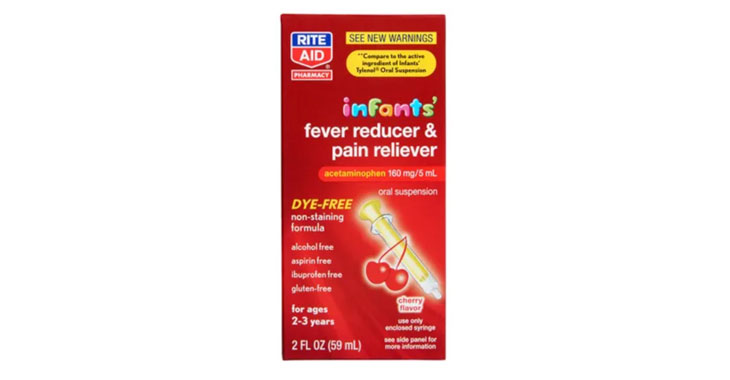
Allegations: Marketing products as safe pain relievers for pregnant women without warning consumers that scientific evidence shows prenatal exposure to APAP can cause neurodevelopmental disorders in children
Allegations: Misleadingly marketing products as “Maximum Strength” when there are other products that contain more lidocaine
Allegations: Failing to disclose that products may contain a dangerous substance that increases the risk of serious adverse health consequences and death
Allegations: Failing to disclose that products contain a harmful substance and may increase the risk of contracting invasive infections
An FDA panel’s recent findings has led to a flood of lawsuits.
Lawsuits claim infant-specific products aren’t any different than acetaminophen medications for older children.
Drugstore chain’s No. 1 doctor-recommended claims come down in wake of NAD inquiry.
These claims are tough to swallow.
Unproven cold prevention and treatment claims are nothing to sneeze at.