CATrends: Fake Discounts on Fitness Products
When the sale price is the regular price.
Information about federal false advertising class actions filed around the country. Please note, our list is neither comprehensive nor reflective of TINA.org’s opinion.
When the sale price is the regular price.
Consumers balk at hidden junk fees.
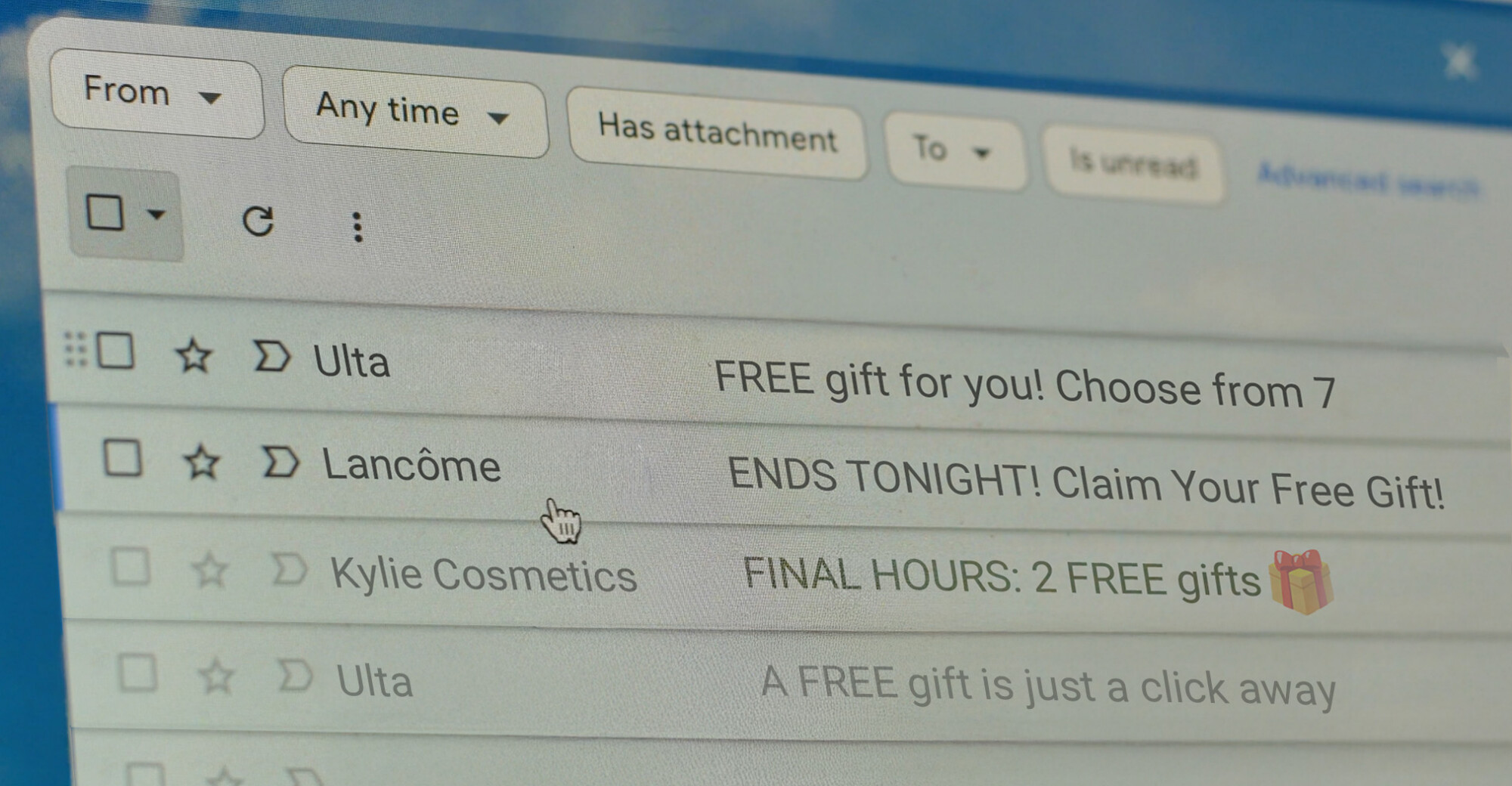
Lawsuits target misleading subject lines.
Allegations: Falsely marketing that supplements are “Natural Brain Enhancer[s]” that support “attention, learning, and memory” when scientific evidence shows that the ingredients in them do not provide the promised results
Allegations: Failing to disclose the terms of subscriptions
Allegations: Falsely marketing products as clinically proven to improve mental clarity and concentration, alleviate stress and tension, and promote a positive mindset
Allegations: Falsely marketing that supplements support “memory, focus, and cognitive processing speed” when scientific evidence shows the ingredients in them do not provide such results
Allegations: Falsely marketing that it has clinical support for claims that products improve memory, focus and mental processing speed
False advertising class-action lawsuits filed against Quincy Bioscience regarding its marketing of Prevagen