Bespoke Apothecary
Products marketed to treat COVID are pulled in wake of FDA/FTC warning letter.
September 2013: Plaintiffs voluntarily dismissed this lawsuit against GNC. The reasons for the dismissal have not yet been disclosed.
July 2013: A class-action lawsuit was filed against GNC for allegedly deceptively marketing its TriFlex supplements. The plaintiffs allege that GNC markets TriFlex, which consists primarily of glucosamine hydrochloride and chondroitin sulfate, as a supplement that will help promote mobility and flexibility, improve joint comfort, and cushion joints, without adequate scientific support for those claims. (Distefano-Presutti et al. v. GNC Corp., Case No. 13-cv-01100, W.D. Penn.)
For more information about other class-action lawsuits regarding glucosamine and TINA.org’s coverage of the issue, click here.
Products marketed to treat COVID are pulled in wake of FDA/FTC warning letter.
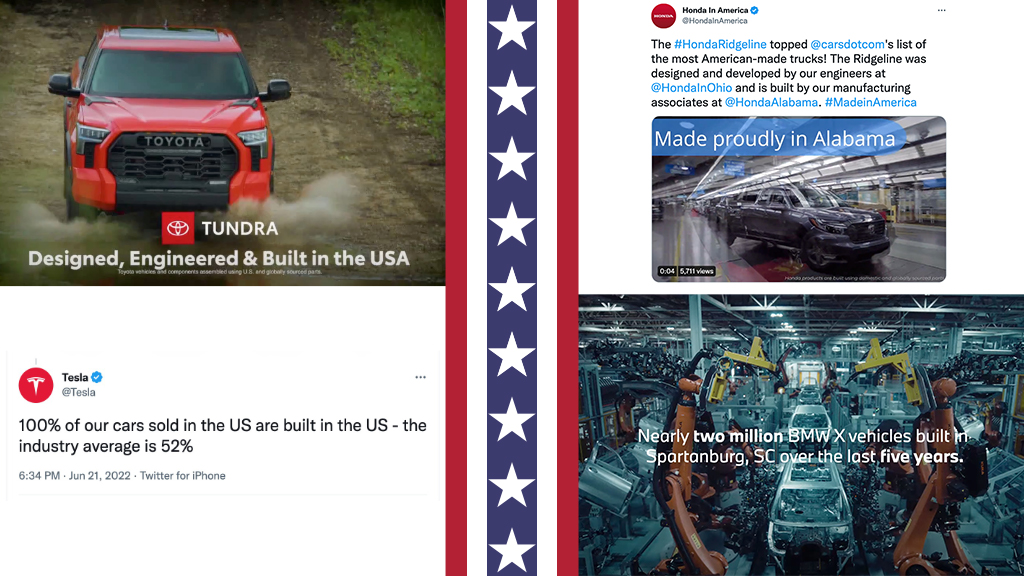
MADISON, CONN. November 7, 2022 – No Super Bowl is complete without at least one car commercial teeming with red, white and blue flags and blatant nods to Americana with…
Jury puts a dollar value on a competitor’s false U.S.-origin claims.
Not according to the cooking instructions on the side of the box.
TINA.org puts the auto industry on notice.