ChapStick Natural Claims
Allegations: False natural claims
Wong et al. v. GlaxoSmithKline Consumer Healthcare Holdings (US) LLC
24-cv-943, S.D. Cal.
(May 2024)
Emergen-C dietary supplements
Misleadingly marketing that products are made with natural flavors when they contain an artificial flavoring ingredient (malic acid)
Pending
Allegations: False natural claims
Allegations: Falsely marketing products as containing “Natural Flavors”
Allegations: Falsely marketing medicine as “Non-Drowsy” when the active ingredient causes drowsiness
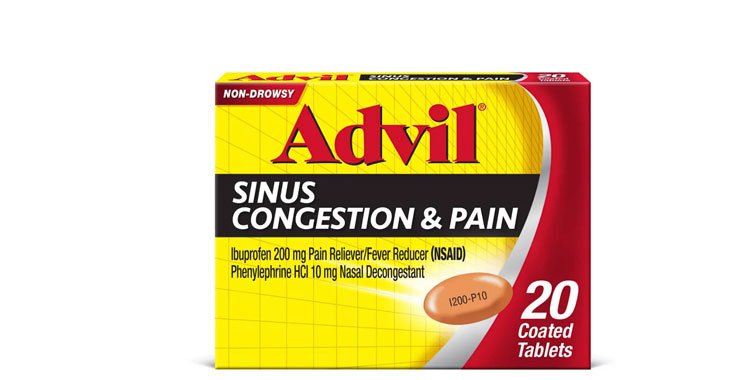
Allegations: Falsely marketing that medicines treat nasal congestion
Allegations: Misleadingly marketing the ingredients in and the health benefits provided by products
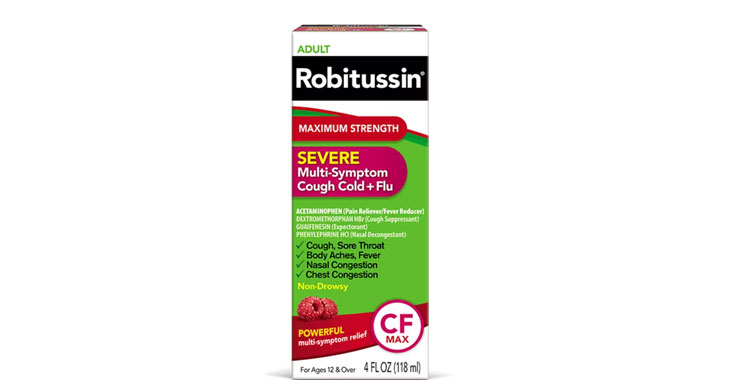
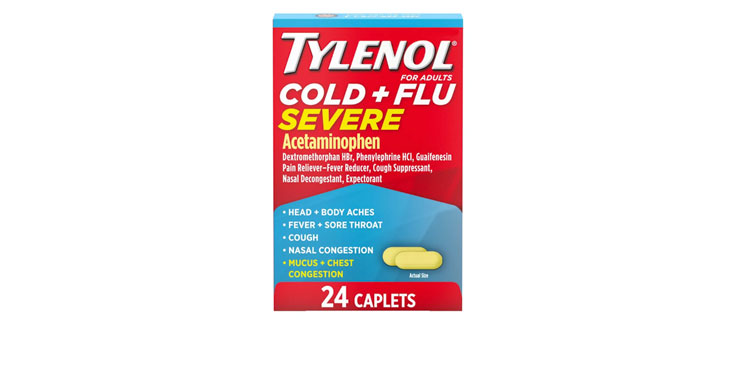
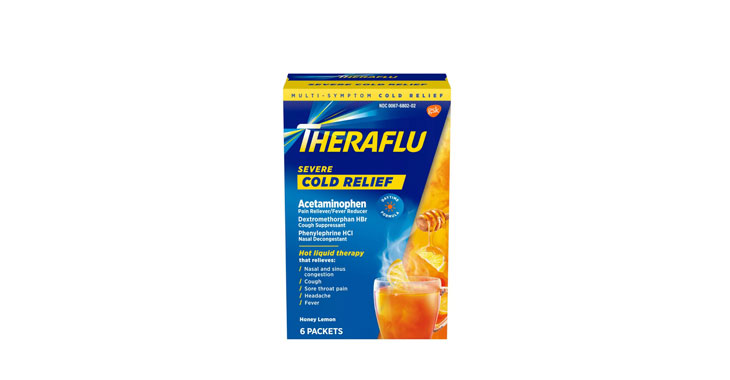
Allegations: Falsely marketing medicines as decongestants
Allegations: Falsely marketing that medicines treat congestion
Allegations: Falsely marketing medicines as nasal decongestants
Allegations: Falsely marketing medicines as nasal decongestants
Allegations: Falsely marketing medicines as decongestants
Allegations: Falsely marketing products as decongestants
Allegations: False natural claims
Allegations: Falsely marketing products as non-drowsy when an ingredient in them causes drowsiness
Allegations: Misleadingly marketing Abreva cold sore treatments
Several products marketed as ‘non-drowsy’ contain an ingredient that causes drowsiness, lawsuits claim.
Lip balm’s own directions seem to contradict its “8 Hour Moisture” labeling claim.
Cue the play-off music.
Is Big Pharma marketing a drug to help aging men with low sex drives or really selling a made-up disease?
GSK will pay out $105 million in off-label use case.