UNBS CBD Gummies
Spam email leads to a fake endorsement from Dr. Oz, among other celebrities.
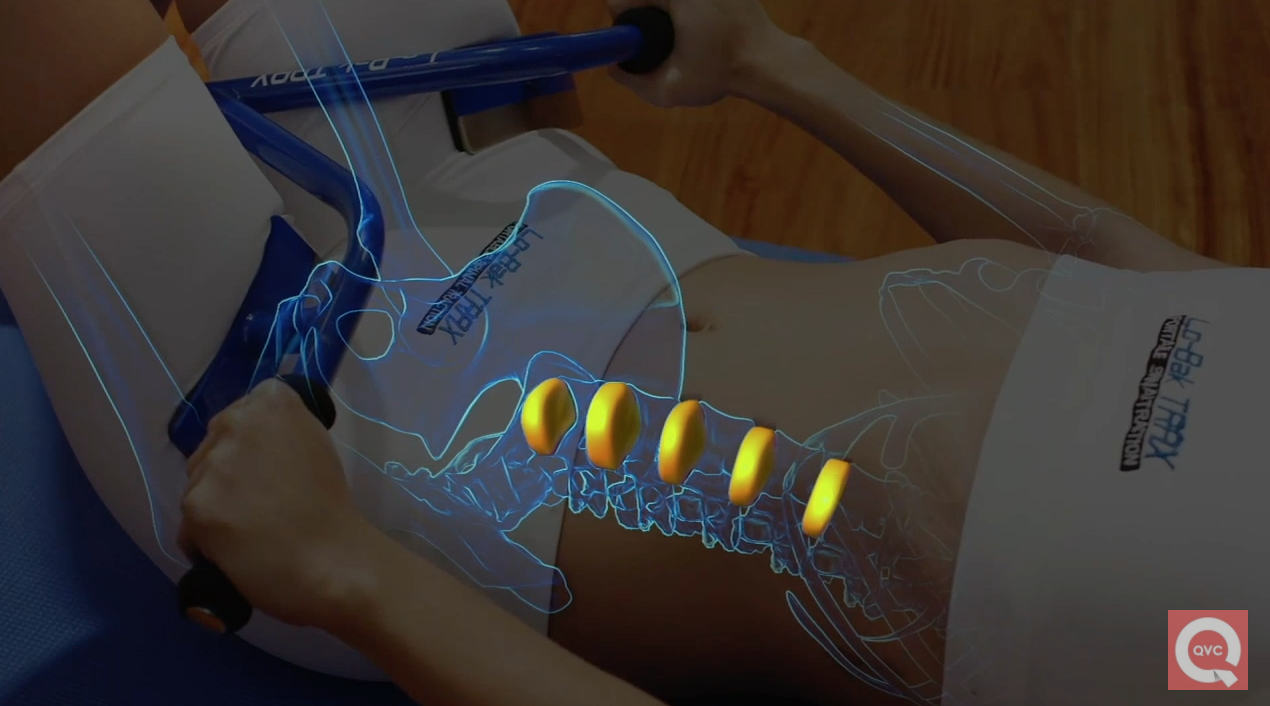
The Lo-Bak TRAX may look like a pair of handlebars — and sound like a child’s mountain bike of the 90s — but, according to the device’s marketers, it’s capable of helping alleviate pain associated with a whole host of back problems.
In fact, the $40 device, which you push against the top of your thighs while lying down, is “FDA cleared” for spinal degenerative joint disease, herniated discs, muscle spasms, spinal stenosis, sciatica … “you name it,” says the host of this QVC commercial for the product, who claims that more than 250,000 units have been sold. The product’s founder, Lori Greiner of Shark Tank fame, and its creator, Dr. Roland Berthiaume, also tout its FDA clearance for these conditions in the segment, with the latter saying it validates the Lo-Bak TRAX.
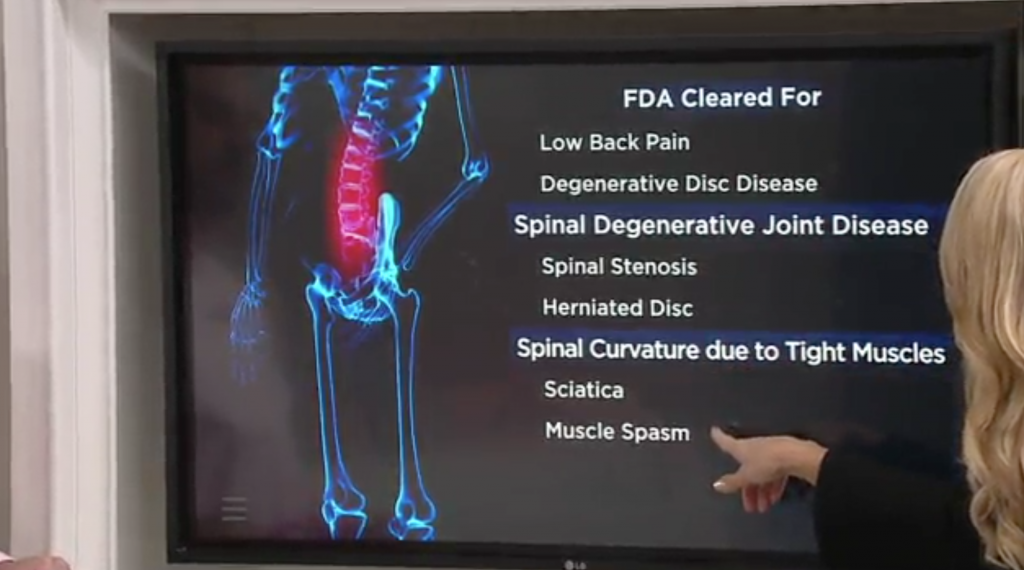
But what the product’s marketers may want you to think “FDA cleared” means and what it actually means may be two different things. That’s because there’s a big difference between “FDA cleared” and “FDA approved.” The truth is much more is required of marketers whose medical devices must be “approved” by the FDA. Among other things, they are subject to more rigorous testing than those seeking “cleared” status and they “must provide reasonable assurance of the device’s safety and effectiveness,” says the FDA.
Marketers who say their device has been “FDA cleared,” on the other hand, have only proven to the FDA that their thing is “substantially equivalent” to a thing that has already been deemed safe and effective by the agency for the same use. For Lo-Bak TRAX, that thing is the Teeter Hang Up’s Inversion Table, which, at this point you may not be surprised to hear, makes similar “FDA cleared” claims on its own website.
Read more about the differences between FDA-cleared and FDA-approved medical devices here.
Our Ad Alerts are not just about false and deceptive marketing issues, but may also be about ads that, although not necessarily deceptive, should be viewed with caution. Ad Alerts can also be about single issues and may not include a comprehensive list of all marketing issues relating to the brand discussed.
Spam email leads to a fake endorsement from Dr. Oz, among other celebrities.
Fake Business Insider reporter Brandt River strikes again.
Snake venom for the win?